Linking ks to surface coverage |

|

|
|
Linking ks to surface coverage |

|

|
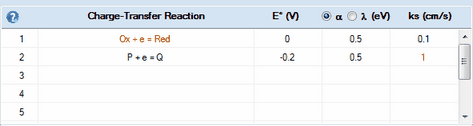
Let's assume there are two (or more) species undergoing charge transfer reactions
Ox + e = Red
P + e = Q
Let's also assume that the species involved in the first redox couple (written in red) are (strongly) adsorbed on the electrode surface. In such a situation the second charge-transfer process may proceed through the covered and uncovered part of the electrode with a different rate constant. This can be simulated by DigiElch by linking the heterogeneous rate constant of the second CT-reaction to the surface coverage of the species involved in the first redox couple:
1.Click on the Edit command in the Tabbed Window: Simulations and enter the above charge transfer reactions. Activate the Enable Adsorption check box for the first one.
2.Enter values for the standard potential, E° (V), and the heterogeneous rate constant, ks, for both reactions. In the case of the second CT-reaction the value of ks should refer to the charge transfer occurring through the uncovered part of the electrode.
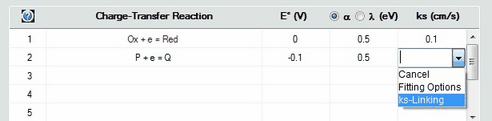
3.Then click with the right mouse button on the ks-value that is to be linked with the surface coverage of another species. Select ks-Linking in the appearing Combo box as shown in the following picture
4.Use the Combo box in the appearing dialog box to select the CT-reaction containing the species to which surface coverage the ks - value is supposed to be linked. In the actual example only one CT-reaction comprising adsorbed species is present.
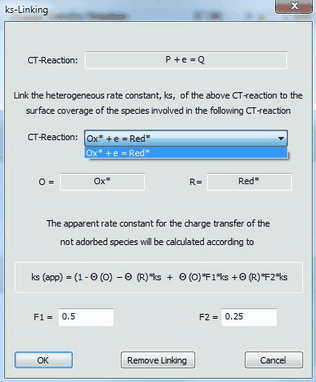
5.Enter values for the factors F1 and F2 and finish with OK.
When proceeding in this way, the second charge transfer process occurs with the standard rate constant, ks, through the uncovered part of the electrode while it proceeds with a standard rate constant ks' = F1*ks and ks'' = F2*ks
through the parts of the electrode covered by the species "Ox" and "Red", respectively. Heterogeneous rate constants which are linked to the surface coverage of an adsorbed species are plotted in red.